|
This applies to everything, not just the behaviour of gases. For this reason (and sometimes others), distant sounds are not only less loud, but also less bright than close ones.Work is equal to force times distance. This diffusion takes mechanical energy out of the wave, especially at high frequencies, and the effect increases with transmission distance. So this process is less negligible for high frequencies, because the shorter half wavelength more than compensates for the shorter half period. However, the time taken for heat to diffuse goes as the square of the distance. Of course in half a period of a sound wave, there is not much time for heat to diffuse one half wavelength. So we expect that some heat will diffuse to the neighbouring pressure minima. When does this fail? At the pressure maxima in a sound wave, the temperature is high. We mentioned above that 'sound transmission in air is close to adiabatic for normal frequencies and transmission distances'. Of course, we may now substitute into (vi) from the equation of state (ii) to see how P and T or T and V are related for an adiabatic process. At constant pressure, you need to put in not only the heat that raises the internal energy, but also an amount of heat equal to the work done when the system undergoes thermal expansion. On reflection, you'll agree that c P > c V: At constant volume, all the heat you put in goes to increasing the internal energy and thus raising the temperature. Hence for an adiabatic process in an ideal gas, So we can write the preceding equation as We know that d( ln x) = dx/x (see calculus for a proof). Let's not stop here, however, because we are only a few lines away from an important result. This is the main result that we need to discuss sound waves: we have just shown that the proportional change in pressure is – γ times the proportional change in volume or γ times the proportional change in density. The ratio of specific heats is given a standard symbol. So the specific heat of an ideal gas at constant pressure is just c P = c v + R. So, if we add heat to a gas at constant pressure, we have to extra heat RdT for each mole of gas, beyond the heat we should have to add at constant volume. From the equation of state of an ideal gas (ii), we can calculate the work dW done at constant pressure: it is just PdV = nRdT. Now if the gas is ideal, its internal energy is all kinetic, so the internal energy U depends only on T. (i) and (ii) give respectively these expressions for nc v dT: We may now combine equations (i) and (iii) to eliminate T. Now the equation of state of an ideal gas is So it follows that definition that the specific heat at constant volume is, where n is the number of moles. So the added heat increases the internal energy U. If we add heat at constant volume, then the gas does not expand and so doesn't do work. The first term may be related to the specific heat, which is defined as the heat added per unit temperature change per mole of substance. The second term is easy: the work done dW when a system changes its volume V by dV is PdV. Substituting this in the first law and rearranging gives The first law of thermodynamics relates the change in internal energy dU to the work dW done by a system and the heat dQ added to it.īy definition, in an adiabatic process, the heat exchanged dQ = 0. 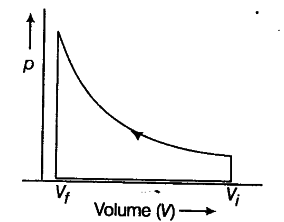
Air at atmospheric pressure is a nearly ideal gas, and sound transmission in air is close to adiabatic for normal frequencies and transmission distances. This page is an appendix to our discussion of sound. How does pressure vary with volume change during adiabatic expansion and compression in an ideal gas?
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
